Another Coronavirus Emerges: U.S. Domestic Response to 2019-nCoV
The Emergence of 2019-nCoV
On December 31, 2019, the World Health Organization (WHO) was informed of a cluster of pneumonia cases in Wuhan, China. Illnesses have since been linked to a previously unidentified strain of coronavirus, designated 2019 novel Coronavirus, or 2019-nCoV. To date, thousands have been infected, mostly in China, and over 100 have died. The disease has spread to several other countries, including the United States. As the scope of the epidemic widened in China, the U.S. Centers for Disease Control and Prevention (CDC) stated on January 27, 2020, that "the immediate health risk from the new virus to the general American public is low currently." With the situation rapidly changing, both WHO and CDC post frequent updates.
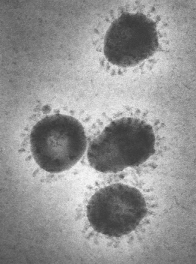
Coronaviruses (see Figure 1) are common respiratory pathogens, usually causing mild illnesses such as the common cold. The global health community is closely monitoring 2019-nCoV because of the severity of symptoms (including death) among those infected, and the speed of its spread worldwide. 2019-nCoV is causing the third-serious novel coronavirus outbreak in modern times, following severe acute respiratory syndrome (SARS) in 2002 and Middle East Respiratory Syndrome (MERS) in 2012. Experts do not know the origin of 2019-nCoV, though genetic analysis and other features suggest an animal source.
 |
|
Source: CDC Public Health Image Library: https://phil.cdc.gov/Details.aspx?pid=10270. Note: Surface projections on the viral envelope of a poultry coronavirus form the halo or "corona" that gives this virus group its name. |
Infection and Transmission
Health officials and researchers are still learning about 2019-nCoV. According to CDC, 2019-nCoV typically causes respiratory infections characterized by a fever, cough, and sometimes breathing difficulty—a suite of symptoms that is common during influenza season. Features that have yet to be clarified include routes of transmission (e.g., through the air, from contaminated surfaces), the incubation period (the time between infection and the onset of symptoms), and whether an infected person without symptoms can transmit infection. Although it was first thought that all 2019-nCoV infections resulted from an animal contact, Chinese officials recently reported evidence of person-to-person transmission.
The U.S. Domestic Response
In the United States, communicable disease control involves collaboration among federal agencies, state health departments, and international partners. Authority to compel isolation (for sick patients), quarantine (for healthy exposed persons), and disease reporting generally rest in state law. The Secretary of Health and Human Services (HHS) and, by delegation, CDC have broad authority to assist in the control of communicable diseases through international cooperation, federal-state cooperation, and public health emergency response activities. In addition, CDC (by delegation) has explicit authority to detain, examine, and release persons arriving into the United States, and traveling between states, who are suspected of having a communicable disease.
Case Identification and Investigation
CDC has developed a diagnostic test for the 2019-nCoV virus, but it is not widely available. At this time, all testing is being done by CDC, which has announced plans to share the test with domestic and international public health partners. Until local testing is available, CDC urges clinicians to consider a patient's travel history and a 14-day incubation period in deciding whether to obtain patient specimens (e.g., blood, urine) for testing. Health care providers are urged to report possible cases to state and local health departments, which then report to CDC. CDC is facilitating the collection and transport of specimens.
Although CDC reports that no person-to-person transmission has been confirmed in the United States, disease investigation for confirmed cases involves contact tracing—identifying and evaluating individuals who had contact with the patient to determine if transmission has occurred. These investigations are generally conducted by state and local health officials.
Clinical Guidance
No vaccine or specific treatment exists for 2019-nCoV infection at this time. Based on knowledge of the novel virus and other coronaviruses, CDC has developed guidance for isolation and other precautions for patients under investigation or receiving supportive care for 2019-nCoV infection. A key goal is the prevention of disease transmission to health care workers, who may be exposed through high-risk procedures such as maintaining an ill patient on a ventilator.
Travel Notices and Entry Screening
CDC and the State Department have issued advisories to reconsider or avoid unnecessary travel to China, including a rare Level 4 advisory from the State Department to avoid all travel to Hubei Province. In collaboration with U.S. Customs and Border Protection, CDC is screening passengers returning on flights from certain areas of China at 20 U.S. airports, looking for symptoms of illness and disseminating information. On January 29, a U.S. government-chartered flight from Wuhan, China, repatriated 201 Americans, who have been screened for illness and advised on procedures to follow should symptoms arise in the coming weeks.
Medical Countermeasures
CDC and the National Institutes of Health (NIH) continue to study the virus to inform response efforts. NIH reports that a vaccine candidate is in development and could be available for initial clinical trials within three months.
Considerations for Congress
CDC and other federal agencies may have the authority needed to respond to the 2019-nCoV epidemic, but they may lack available funds to support their efforts. Annual appropriations and standing transfer authority provide the HHS Secretary with some flexibility. Under certain circumstances the Secretary may access a Public Health Emergency Fund, but the fund does not have an available balance at this time. Generally, funding has not been available under the Robert T. Stafford Disaster Relief and Emergency Assistance Act (the Stafford Act) for the response to infectious disease outbreaks. As a result, Congress provided supplemental appropriations for the response to the recent Ebola and Zika virus outbreaks.
Subsequently in 2018, Congress established an Infectious Disease Rapid Response Reserve Fund (IDRRRF) for CDC, providing $50 million for FY2019 (P.L. 115-245) and $85 million for FY2020 (P.L. 116-94), available until expended. These funds can be made available for an infectious disease emergency if the HHS Secretary either (1) declares a Public Health Emergency or (2) determines that the infectious disease outbreak has significant potential to occur and, if it occurs, the potential to affect national security or the health and security of U.S. citizens, both domestically and abroad. HHS Secretary Alex Azar has not declared the 2019-nCoV outbreak to be a Public Health Emergency, but has issued a determination allowing the allotment of $105 million from the IDRRRF for the 2019-nCoV response.
The trajectory of the 2019-nCoV outbreak is unknown at this time. Congress may choose to conduct oversight of federal response efforts, and monitor the expenditure of funds, as the situation progresses.